IQVIA’s Global Trends in R&D 2026 confirms what the digital health data has been signalling for two years: pharma’s productivity problem lives in execution, not discovery — and the capital is already reallocating.
KEY TAKEAWAYS
- Health Management Solutions captured $5.6 billion in 2025 venture funding, representing 18.8% of all global digital health funding.
- AI has become the indisputable core infrastructure: an unprecedented 83% of HMS capital flowed into AI-powered ventures by early 2026, up from 61% in 2021.
- The market is rapidly consolidating around late-stage platforms — average deal size tripled from $11.9 million in Q1 2022 to $35.4 million in Q1 2026.
- Healthcare Operations & Workflow captures 46% of health-system partnerships, proving systems are prioritising measurable ROI and capacity relief.
- Asia Pacific is the most asymmetric growth opportunity globally with +87% year-on-year funding growth in 2025; the Middle East +54%; North America plateaued at −5% and Europe contracted −15%.
- WHOOP closed a $575 million Series G and Harbinger Health a $100 million Series B1 — capital is concentrating in execution-focused, AI-native platforms.
1. What does the IQVIA 2026 report tell us about R&D productivity?
The IQVIA Institute’s Global Trends in R&D 2026 is, at first pass, a measured document. R&D funding levels held up in 2025 but were below 2024 levels. Deal activity between pharma companies rose, with deals between China-headquartered and international companies reaching an all-time high. Seventy-nine novel active substances (NAS) launched globally in 2025 — of which thirty were global first-in-class — bringing the five-year total to 388 (or 369 excluding COVID-19 vaccines and therapeutics). A further 30 NAS were launched only in China, never reaching global markets.
The uncomfortable numbers sit in the productivity section. End-to-end clinical development timelines grew. Inter-trial intervals lengthened by three months. Oncology enrolment moved faster, but because oncology now dominates the trial mix, overall durations still rose. The improvements pharma had clawed back post-pandemic have quietly reversed. IQVIA points to AI as the most credible lever to bend the curve, noting materially higher success rates among AI-driven programmes.
That is a sober diagnosis. It is also, read from the outside, incomplete. The report is written from the inside of pharma looking out. The more revealing question is what is happening in the ecosystem that supplies pharma with its execution infrastructure. That ecosystem is digital health, and the data on it tells a sharper, more actionable story.
2. The bottleneck has shifted to execution — and the data proves it
Galen Growth has been making this case since early 2026: the bottleneck in bringing new medicines to patients is no longer scientific discovery; it is clinical execution. The industry has taught machines to design complex molecules in minutes, yet it still relies on deeply analogue methods to identify the human beings required to test them.
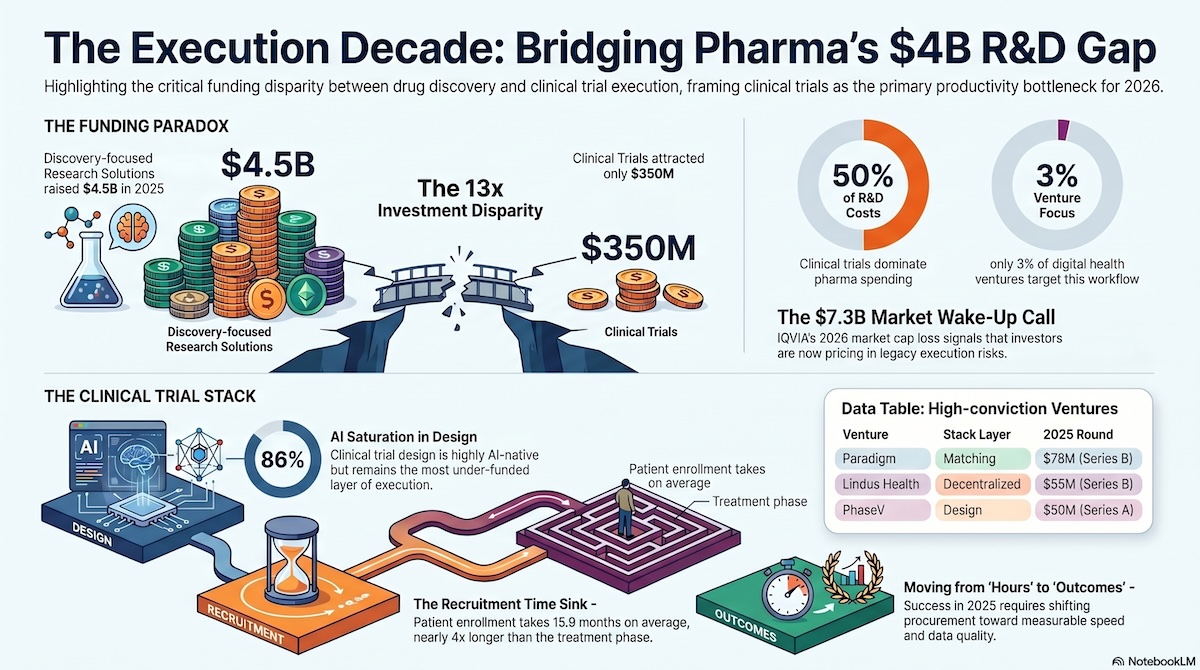
The financial asymmetry is stark. Clinical trials consume more than 50% of total pharmaceutical R&D costs. Yet, according to HealthTech Alpha, only around 3% of digital health ventures focus on the clinical trial workflow. Between 2020 and 2025, TechBio (discovery-focused) funding surged by 75% to $4.8bn, while funding for clinical trial innovation contracted by 30%, with just $1.6bn deployed in 2025. Capital has been pouring into the front of the funnel, while the biggest cost centre in the pipeline has been systematically underfunded.
That mismatch is precisely what the IQVIA 2026 cycle-time numbers reflect. Lengthening trial durations is not a mystery. They are what happens when the discovery layer accelerates while the execution layer stalls. The molecules are ready. The protocols and patients are not, or rather, they cannot be, found, enrolled, and retained quickly enough to match the throughput that discovery engines are now capable of producing.
3. Where the money actually went in 2025 — funding and exits
HealthTech Alpha tracks the full global digital health venture base — 16,210 ventures at the time of writing, mapped through a proprietary ontology across categories, clusters, therapeutic focus, technologies, and target users. Looking at 2025 capital flow through that lens, and separating primary funding from exits, clarifies what the IQVIA narrative only hints at.
Global digital health recorded 1,128 venture equity rounds totalling $25.7bn in 2025 (Seed through Series G, inclusive of strategic, convertible and bridge rounds; average round ~$23m), plus 209 exit transactions worth $15.0bn (M&A, IPO, SPAC and Pre-IPO combined) and a further $4.9bn across 293 transactions in non-equity capital — grants, debt financing, private equity and post-IPO equity. The exits line did real work: 194 M&A transactions delivered $9.7bn, IPOs added $3.5bn across just 12 listings, and a single SPAC contributed $1.5bn. Capital is not retreating from digital health — it is concentrating, and the exits market is where the concentration is most visible.
A note on definitions: this analysis uses a strict venture-equity definition of “primary funding” to align with how pharma strategists, corporate development teams, and venture investors typically interpret the data. Grants, debt financing, post-IPO equity and private equity buyouts are reported separately. Under a broader non-exit definition that includes all of these, the 2025 total reaches $30.6bn across 1,422 transactions.
Research Solutions: where the capital genuinely piled in
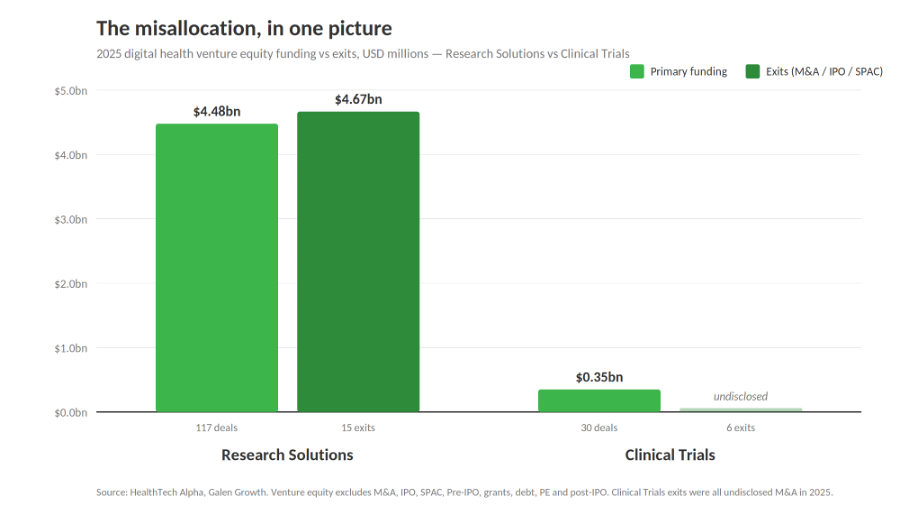
The Research Solutions cluster — the part of digital health that directly supplies pharma R&D with AI discovery engines, bioinformatics, and omics platforms — absorbed $4.5bn in venture equity across 117 rounds and $4.7bn across 15 exit transactions in 2025, for a total capital footprint of roughly $9.2bn. That cluster accounts for just 854 of 16,210 tracked ventures (~5% of the ecosystem), yet absorbed more combined capital than any other cluster. Structurally under-represented, strategically over-weighted.
Within Research Solutions on a venture equity basis, Drug Discovery dominated, with 69 rounds totalling $2.8bn, at an average round size of $46m. Bioinformatics added $1.1bn across 28 rounds, at an average of $43m per round. Omics-related research raised $0.5bn across 20 rounds, with an average of $36m per round. The exits line amplified this: Drug Discovery alone delivered $3.3bn across six transactions (led by Nanyang Biologics’ $1.5bn SPAC and Orbital Therapeutics’ $1.5bn M&A), with a further $0.9bn from the Rznomics IPO in Omics and $0.5bn from the Caris Life Sciences IPO in Bioinformatics. These are not seed cheques. They are infrastructure commitments, and the average venture equity round size is several times the ecosystem mean.
The named rounds make the point concrete. Largest 2025 primary rounds in Research Solutions on HealthTech Alpha: Isomorphic Labs (UK) $600m strategic, Pathos (US) $365m Series D, Truveta (US) $320m Series C, Mirador Therapeutics (US) $250m Series B, Valo Health (US) $190m strategic, SandboxAQ (US) $150m Series E, and Enveda (US) $150m Series D. Largest 2025 exits: Nanyang Biologics (Singapore) $1.5bn SPAC, Orbital Therapeutics (US) $1.5bn M&A, Rznomics (South Korea) $848m IPO, Caris Life Sciences (US) $494m IPO, and Insilico Medicine (Hong Kong) $292m pre-IPO, with further strategic M&A on Fabric Genomics, Akoya Biosciences, and Target RWE. The exits list alone does most of the work of explaining why strategic buyers now view Research Solutions as a core infrastructure category rather than a speculative one.
Geographically, Research Solutions venture equity in 2025 skewed heavily to North America ($2.8bn, 69 rounds, 60 ventures), followed by Europe ($1.2bn, 28 rounds, 28 ventures) and APAC ($0.4bn, 17 rounds, 14 ventures). At the exit end of the capital stack, APAC punches far above its venture-round weight — the Nanyang Biologics SPAC and the Rznomics IPO alone account for roughly $2.3bn of regional capital absorbed. The China-linked cross-border deal activity IQVIA flagged as reaching an all-time high is also visible in the HealthTech Alpha signal, particularly at the exit end.
Clinical Trials: the paradox sharpens
The Clinical Trials cluster tells a very different — and more urgent — story. In 2025, it attracted only $350m in venture equity across 30 rounds, with an average round size of just $12m. That is barely above one per cent of total digital health venture equity. For the cluster that targets the R&D cost centre, IQVIA has just confirmed it is getting slower, not faster. Six exits were recorded in the cluster, all M&A, none with publicly disclosed values — a quiet wave of consolidation rather than a splashy liquidity event.
The distribution across the Clinical Trial Stack is instructive. Decentralised Clinical Trials led the cluster at $122m across 9 venture equity rounds. Clinical Trial Design followed at $97m across 12 rounds. Clinical Trial Matching took $93m across just 5 rounds. Data Collection Tools attracted $39m across 4 rounds. The design layer is the most AI-saturated (86% of ventures use AI, per HealthTech Alpha) yet still among the lowest-funded in absolute dollars — a textbook supply-and-demand tension for disciplined investors.
The 2025 named rounds capture where the pockets of conviction actually sit: Paradigm (US, Clinical Trial Matching) $78m Series B; Lindus Health (UK, Decentralised Clinical Trials) $55m Series B; PhaseV (Israel, Clinical Trial Design) $50m Series A; DIP (Singapore, Decentralised Clinical Trials) $50m Series D; Motivity (US, Data Collection Tools) $27m Series B. On the exits side, M&A activity touched Deep 6 AI and Syapse (Matching), Circuit Clinical, MedNet Solutions and Mondosano (Decentralised), and Real Life Sciences (Data Collection) — a clean read-through that strategic buyers are picking up proven execution assets even when venture capital is distracted.
The contrast with Research Solutions is the whole point. The cluster that fixes the productivity problem IQVIA just diagnosed is attracting roughly one-thirteenth the venture equity of the cluster that feeds the discovery engine — and within that, Decentralised Clinical Trials, the layer the public markets have already validated through M&A, is taking just $122m of new risk capital globally. That is the misallocation to correct in 2026.
4. The Clinical Trial Stack: where execution value concentrates
To understand where value is actually being created — and where it is about to be captured — the clinical execution layer needs to be decomposed rather than treated as a single category. Galen Growth frames this as the Clinical Trial Stack: four layers, each with distinct economics and distinct winners.
“The industry has taught machines to design complex molecules in minutes. It still relies on deeply analogue methods to identify the human beings required to test them. That asymmetry is the productivity story of the next decade.”
— Julien de Salaberry, Founder & CEO, Galen Growth
Design
Protocol formulation, feasibility, endpoint structuring. The most AI-saturated layer of the stack: 86% of ventures in clinical trial design actively use AI tools, according to HealthTech Alpha. Also, the most under-represented by venture volume — just 14% of the stack — which signals a clear supply-demand tension. Names to watch: Phesi (Alpha Score 60.2) and Risklick (47.3).
Matching
Patient identification, eligibility, reachability, enrolment, retention. Citeline 2024 data pegs enrolment alone at an average of 15.9 months, more than four times the 3.9-month treatment phase. The time sink in clinical development is finding and enrolling patients, full stop. Leaders on the HealthTech Alpha signal include Inato (Alpha Score 64.0) and uMed (Partnership Score 85.2).
Decentralised execution
Hybrid and virtual trial architectures are now the default operating model, not an experiment. The financial markets have already voted: 59% of clinical trial venture M&A between 2021 and 2025 was in the Decentralised Clinical Trials category. Medable (Alpha Score 81.6, Partnership Score 89.5) sits at the top of that cohort.
Data collection
Continuous capture via wearables, remote sensors, and audit-grade platforms. Technology alone does not create a moat here; Good Clinical Practice-grade controls do. In regulated evidence, trust is not a feature; it is the product. Qureight (Partnership Score 84.1) is a clean example of how defensibility is built through data integrity, not interface design.
| Venture | Stack Layer | Country | 2025 Round | Stage |
| Paradigm | Matching | US | $78m | Series B |
| Lindus Health | Decentralised | UK | $55m | Series B |
| PhaseV | Design | Israel | $50m | Series A |
| DIP | Decentralised | Singapore | $50m | Series D |
| Motivity | Data Collection | US | $27m | Series B |
| Deep 6 AI | Matching | US | undisclosed | M&A (exit) |
| Syapse | Matching | US | undisclosed | M&A (exit) |
| Circuit Clinical | Decentralised | US | undisclosed | M&A (exit) |
| MedNet Solutions | Decentralised | US | undisclosed | M&A (exit) |
| Mondosano | Decentralised | Germany | undisclosed | M&A (exit) |
| Real Life Sciences | Data Collection | US | undisclosed | M&A (exit) |
5. The CRO repricing is already underway
The public market read on this shift is already brutal. In early 2026, IQVIA lost $7.3bn in market capitalisation on a single guidance miss — not because its Q4 was weak, but because its 2026 profitability outlook was. Investors are pricing in a hard reality for traditional contract research: AI-driven margin compression, a mandatory “profitability valley” as incumbents fund their own internal AI and cloud build-outs, and accelerating contract cancellations as sponsors move away from legacy trial models.
This is not a theoretical disruption scenario. It is a market that has decided the service-heavy, human-in-the-loop CRO model is structurally mispriced for an AI-enabled future. The implication for pharma procurement is immediate: the industry will increasingly buy outcomes — measured in recruitment speed, amendment rates, and data quality — rather than billable hours.
6. Pilot purgatory: the real ceiling on scale
If capital is being reallocated and the technology exists, why is the IQVIA cycle-time curve still pointing in the wrong direction? The honest answer is that innovation in clinical trials dies in what founders call pilot purgatory. A successful pilot triggers an exhaustive IT security review. That triggers an electronic health record integration that stalls due to a mismatch of incentives among the clinical budget owner, IT, and the corporate sponsor.
The mortality data are sobering. Only around 10% of clinical trial ventures launched in the last five years have reached Series A. Scaling execution technology is, at root, a coordination problem, not a technology problem. The winners will be the players who industrialise interoperability and alignment across sponsors, CROs, and sites — not the ones with the cleverest algorithm. Between 2024 and 2025, more than 80 new strategic partnerships were forged between health systems and clinical trial technology vendors. That is the shape of the answer.
7. The visibility gap — and why it matters now
IQVIA’s report is built on one of the most complete pharma datasets in the world: funding, trial starts, NAS launches, country utilisation, cycle-time benchmarks, and emerging biopharma participation. The digital health infrastructure that increasingly determines whether those pipeline numbers translate into patient impact is measured far less rigorously. Most public tracking is venture-capital native — deal counts, valuations, exit multiples. That is a reasonable view for an investor. It is a dangerously thin view for a Chief R&D Officer, a Chief Medical Officer, a corporate development team, or a regulator.
The productive question — the one IQVIA is implicitly asking on the pharma side and that the market is not systematically asking on the digital health side — is which ventures are producing durable evidence, real partnerships, clinical traction, and regulatory progress, and which are simply raising rounds. Answering it requires the same discipline the industry has long applied to drug pipelines: a structured ontology, consistent definitions, auditable provenance, and the ability to compare like with like across geographies and therapeutic areas.
That is exactly the work HealthTech Alpha was built to do. Every venture in the platform is classified against a proprietary ontology spanning categories, clusters, therapeutic focus, technologies, target users, and value propositions. Every venture is scored across eight dimensions — Maturity (Alpha), Momentum, Market, Innovation, Money, Evidence, Partnership, and Team — with the inputs linked to underlying clinical trials, regulatory approvals, patents, publications, partnerships, and funding records. The point is not to replace the IQVIA view of pharma. The point is to make the layer beneath it legible on the same terms.
8. What this means
| For investors: the gap between cyclical venture starvation in the Clinical Trials cluster ($350m across 30 rounds in 2025) and the 17% M&A exit rate HealthTech Alpha records for the cluster is a generational mispricing. Underwrite data moats, workflow depth and compliance-grade evidence. Expect software margins to replace human-in-the-loop margins — and structure positions accordingly. The most attractive entry points lie in Decentralised Clinical Trials (where the public markets have already validated demand through M&A) and Clinical Trial Design (where 86% of ventures are AI-native, but only $97m of new equity has flowed in). | For pharma sponsors and corporate partners: abandon the pilot-of-the-quarter habit. Commit to an AI-enabled reference architecture. Shift procurement from hours to outcomes — enrolment speed, amendment reduction, data quality — and align sponsor, CRO and site incentives to match. The IQVIA $7.3bn market-cap loss is the public market’s way of telling pharma procurement that the legacy CRO buying pattern is structurally mispriced. The companies that move first in 2026 will materially outrun those that do not. |
| For health systems and providers: the 80+ new strategic partnerships forged between health systems and clinical trial technology vendors in 2024–2025 are the early signal of where the operating model is going. The institutions that build standardised, interoperable data and consent infrastructure now — rather than negotiating each pilot from scratch — will become preferred sites for sponsors and capture a disproportionate share of trial volume as decentralised execution scales. | For digital health ventures: the era of single-feature SaaS pitches is over. Build into sponsor and CRO workflows from day one. Sell measurable lift in cycle time, not feature breadth. Treat validation, security and auditability as core product, not compliance overhead. And remember that interoperability is no longer a differentiator — it is table stakes. The 10% Series A rate for clinical trial ventures launched in the last five years is the proof: scaling execution technology is, at root, a coordination problem, not a technology problem. The IQVIA Institute has given the industry a clear and honest read on the top of the R&D stack. The bottom of the stack — where pharma productivity is, in practice, now won or lost — is where the Galen Growth point of view sits. The tools exist. The science is ready. The capital is already moving. The only remaining constraint is the speed at which the incumbents industrialise what the data has been telling them for two years: the execution decade has begun. |
FAQ
What is the core argument of The Execution Decade?
The bottleneck in bringing new medicines to patients has shifted from scientific discovery to clinical execution. IQVIA’s Global Trends in R&D 2026 shows pharma cycle times lengthening and inter-trial intervals growing by three months, even as AI-driven discovery accelerates. Digital health capital has begun to reallocate toward the execution layer, but not fast enough: in 2025, the Research Solutions cluster, which feeds discovery, absorbed roughly thirteen times the venture equity of the Clinical Trials cluster, which targets execution. Closing that misallocation is the defining industry task over the next 12 to 24 months.
How much did digital health raise in 2025?
Global digital health attracted $25.7bn in venture equity across 1,128 rounds, Seed through late-stage, in 2025, plus $15.0bn across 209 exit transactions, including M&A, IPO, SPAC and Pre-IPO. A further $4.9bn came through 293 non-equity transactions, including grants, debt financing, private equity and post-IPO equity. Total non-exit capital reached $30.6bn across 1,422 transactions. Exits concentrated in 194 M&A transactions worth $9.7bn, 12 IPOs worth $3.5bn, and a single SPAC worth $1.5bn. All figures are from HealthTech Alpha, Galen Growth.
What is the Clinical Trial Stack?
The Clinical Trial Stack is the Galen Growth framework for decomposing clinical execution into four distinct layers, each with its own economics, leaders, and capital dynamics. In 2025, the stack attracted $350m in venture equity across 30 rounds, with Decentralised Clinical Trials leading at $122m and Clinical Trial Matching trailing at $93m.
Clinical Trial Design covers protocol formulation and feasibility. Clinical Trial Matching covers patient identification, eligibility and enrolment. Decentralised Clinical Trials are platforms for executing hybrid and virtual trial architectures. Data Collection Tools cover wearables, remote sensors and audit-grade platforms.
Why did IQVIA lose $7.3bn in market capitalisation in early 2026?
IQVIA’s 2026 guidance miss triggered a ~$7.3bn market capitalisation loss, not because of weak Q4 results, but because the forward profitability outlook disappointed. Public market investors are pricing in a structural shift for traditional contract research: AI-driven margin compression, a mandatory “profitability valley” as CROs fund their own AI and cloud build-out, and accelerating contract cancellations as sponsors migrate to AI-enabled trial platforms. The signal is that the service-heavy, human-in-the-loop CRO model is being repriced for an AI-native future, and pharma procurement is moving from billable hours toward measurable outcomes such as recruitment speed, amendment rates and data quality.
Which digital health ventures raised the largest rounds in 2025?
In Research Solutions, the largest 2025 venture rounds were Isomorphic Labs, UK, with a $600m strategic round; Pathos, US, with a $365m Series D; Truveta, US, with a $320m Series C; Mirador Therapeutics, US, with a $250m Series B; Valo Health, US, with a $190m strategic round; SandboxAQ, US, with a $150m Series E; and Enveda, US, with a $150m Series D. In Clinical Trials, the largest 2025 venture rounds were Paradigm, US, with a $78m Series B; Lindus Health, UK, with a $55m Series B; PhaseV, Israel, with a $50m Series A; DIP, Singapore, with a $50m Series D; and Motivity, US, with a $27m Series B.
What were the largest digital health exits in 2025?
In Research Solutions, the largest 2025 exits were Nanyang Biologics, Singapore, with a $1.5bn SPAC; Orbital Therapeutics, US, with a $1.5bn M&A transaction; Rznomics, South Korea, with an $848m IPO; Caris Life Sciences, US, with a $494m IPO; and Insilico Medicine, Hong Kong, with a $292m pre-IPO round, alongside strategic M&A involving Fabric Genomics, Akoya Biosciences and Target RWE. In the Clinical Trials cluster, six M&A exits were recorded in 2025, all with undisclosed values: Deep 6 AI, Syapse, Circuit Clinical, MedNet Solutions, Mondosano and Real Life Sciences.
What is “pilot purgatory” and why does it matter?
Pilot purgatory is the stage at which a successful clinical trial technology pilot stalls before enterprise-scale deployment. A successful pilot triggers an IT security review, which in turn triggers an electronic health record integration project, which stalls due to misaligned incentives among the clinical budget owner, the IT function and the corporate sponsor. It is the primary reason the IQVIA cycle-time curve keeps pointing in the wrong direction despite abundant available technology. Only around 10% of clinical trial ventures launched in the past five years have reached Series A. The winners will be the players who industrialise interoperability and incentive alignment across sponsors, CROs and sites, not the ones with the cleverest algorithm.
What is HealthTech Alpha, and how was it used in this analysis?
HealthTech Alpha is Galen Growth’s structured digital health intelligence platform. It tracks 16,000+ ventures globally, classifying each against a proprietary ontology that covers categories, clusters, therapeutic focus, technologies, target users and value propositions. Every venture is scored across eight dimensions: Maturity, Momentum, Market, Innovation, Money, Evidence, Partnership and Team. Inputs are linked to underlying clinical trials, regulatory approvals, patents, publications, partnerships and funding records. All venture-level data in this analysis, including deal counts, funding totals, regional splits, named rounds, Alpha Scores and Partnership Scores, is drawn directly from HealthTech Alpha, accessed April 2026.
What should pharma, investors, founders and policy teams do next?
Pharma sponsors and CRO leaders should abandon the pilot-of-the-quarter habit, commit to an AI-enabled reference architecture and shift procurement from hours to outcomes. Investors should underwrite data moats, workflow depth and compliance-grade evidence. Digital health founders should build into sponsor and CRO workflows from day one, treat validation and auditability as core product, and sell measurable cycle-time lift rather than feature breadth. Pharma strategy, corporate development and policy teams should treat visibility at the execution layer as a core competence, not a research afterthought.
What is the single most important statistic from this analysis?
Clinical trials consume more than 50% of total pharmaceutical R&D costs, yet in 2025 the Clinical Trials cluster of digital health attracted just $350m in venture equity — approximately one-thirteenth of the $4.5bn that flowed into the Research Solutions cluster, which feeds discovery. The cost centre is the largest in pharma; the capital reallocation has barely begun; the execution decade is the correction.
Methodology
Data source: HealthTech Alpha by Galen Growth, accessed April 2026. Funding figures cover transactions dated 1 January 2025 to 31 December 2025. “Venture equity” is defined strictly: Seed through Series G, plus Strategic, Convertible, Bridge, Angel and Pre-A rounds. It excludes M&A, IPO, SPAC and Pre-IPO (reported separately as exits) as well as grants, debt financing, post-IPO equity, private equity buyouts, secondaries and crowdfunding (reported separately as other capital). The Research Solutions and Clinical Trials clusters reflect HealthTech Alpha’s proprietary ontology. Some 2025 figures may be understated where deal terms were not publicly disclosed; in particular, all six 2025 M&A exits in the Clinical Trials cluster were undisclosed. Round-level values are aggregated from publicly disclosed transactions and represent the venture’s headquarters region. The IQVIA Institute figures cited (NAS launches, cycle-time deltas, China-linked deals, AI success rates, five-year forecast) are taken from “Global Trends in R&D 2026” published March 2026.
Related Galen Growth analysis
- Digital health funding in Clinical Trials: Why Execution, Not Discovery, is Pharma’s Next Existential Bottleneck — Galen Growth, March 2026
- HealthTech Alpha — global digital health intelligence platform — Galen Growth
- Galen Growth Insights — full archive of digital health analyses — Galen Growth
How to cite this analysis
About Galen Growth
Galen Growth is the digital health intelligence firm behind HealthTech Alpha, the leading ontology-driven platform tracking the global digital health ecosystem. With operating entities in the US, Europe and Asia, we combine large-scale labelled data, auditable GenAI research and explainable analytics to advise pharma, medical device, insurance, health system, investor and startup clients.